ATROTOS
The science behind ATROTOS.
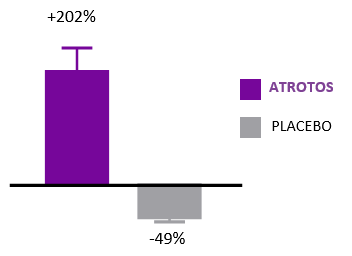
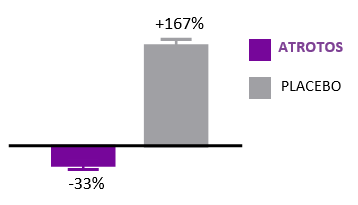
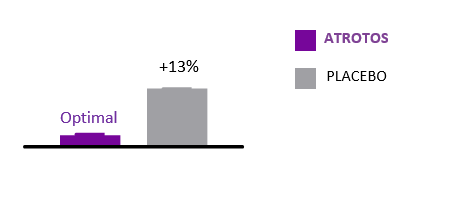
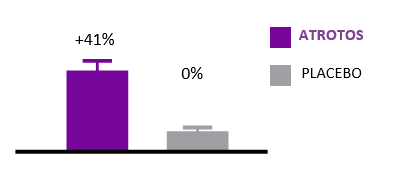
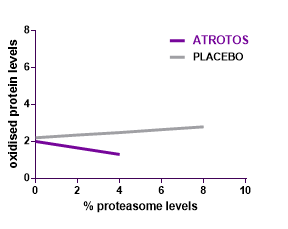
Cellular Senescence
ATROTOS — Senescence Modulation Support
Cellular senescence is a hallmark of biological aging. Senescent cells are metabolically active but non-dividing cells that release pro-inflammatory and tissue-disrupting factors, negatively affecting neighboring healthy cells and contributing to progressive tissue dysfunction.
In controlled in-vitro studies, human fibroblasts exposed to ATROTOS demonstrated extended replicative lifespan compared to untreated controls. These findings indicate a senescence-delaying effect at the cellular level. By supporting healthier cellular aging dynamics, ATROTOS contributes to the maintenance of regenerative potential and functional cellular performance within a structured Healthy Aging & Longevity Platform.
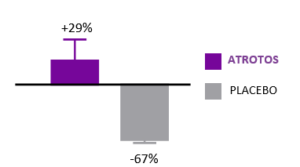
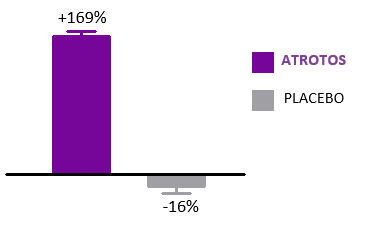
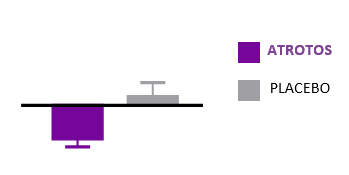
Telomere Dynamics
ATROTOS — Telomere Stability Support
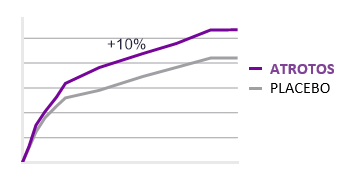
Telomere length is a validated biomarker of cellular aging and replicative capacity. Telomeres function as protective chromosomal end-structures and progressively shorten with each cell division. Accelerated telomere attrition is strongly associated with biological aging and with increased risk burden across multiple age-related conditions.
In controlled in-vitro studies using human fibroblasts, cells treated with ATROTOS did not demonstrate telomere shortening at the point of replicative senescence, compared with untreated controls. These findings support a telomere-stabilizing effect under experimental conditions. Preservation of telomere length is mechanistically aligned with healthier cellular aging trajectories and supports the Healthy Aging & Longevity Platform framework.